Design and synthesis of ONOO − activatable molecular probe with NIR-II

Download scientific diagram | Design and synthesis of ONOO − activatable molecular probe with NIR-II fluorescence. Conditions: (a) Et 2 NH, Pd 2 (dba) 3 , DavePhos, Cs 2 CO 3 , dioxene, 95 °C, 18 h; (b) i CH 3 MgBr, ii HClO 4 ; (c) toluene/n-butanol, 100 °C, 12 h. from publication: Peroxynitrite Activatable NIR-II Fluorescent Molecular Probe for Drug-Induced Hepatotoxicity Monitoring | Drug-induced hepatotoxicity represents an important challenge for safety in drug development. The production of peroxynitrite (ONOO-) is proposed as an early sign in the progression of drug-induced hepatotoxicity. Currently, reported ONOO- probes mainly emit in visible range | Hepatotoxicity, Fluorescence and Drug-Induced Liver Injury | ResearchGate, the professional network for scientists.

Design and synthesis of a small molecular NIR-II chemiluminescence probe for in vivo-activated H2S imaging

A General Strategy for Development of Activatable NIR‐II Fluorescent Probes for In Vivo High‐Contrast Bioimaging - Ren - 2021 - Angewandte Chemie International Edition - Wiley Online Library

Frontiers Recent Progress in NIR-II Contrast Agent for Biological Imaging

Full article: Recent Development in Fluorescent Probes for the Detection of Hg2+ Ions

Crossbreeding” Small-Molecular Weight NIR-II Flavchromenes Endows Activatable Multiplexed In Vivo Imaging

Construction of HPQ-based activatable fluorescent probe for peroxynitrite and its application in ferroptosis and mice model of LPS-induced inflammation - ScienceDirect
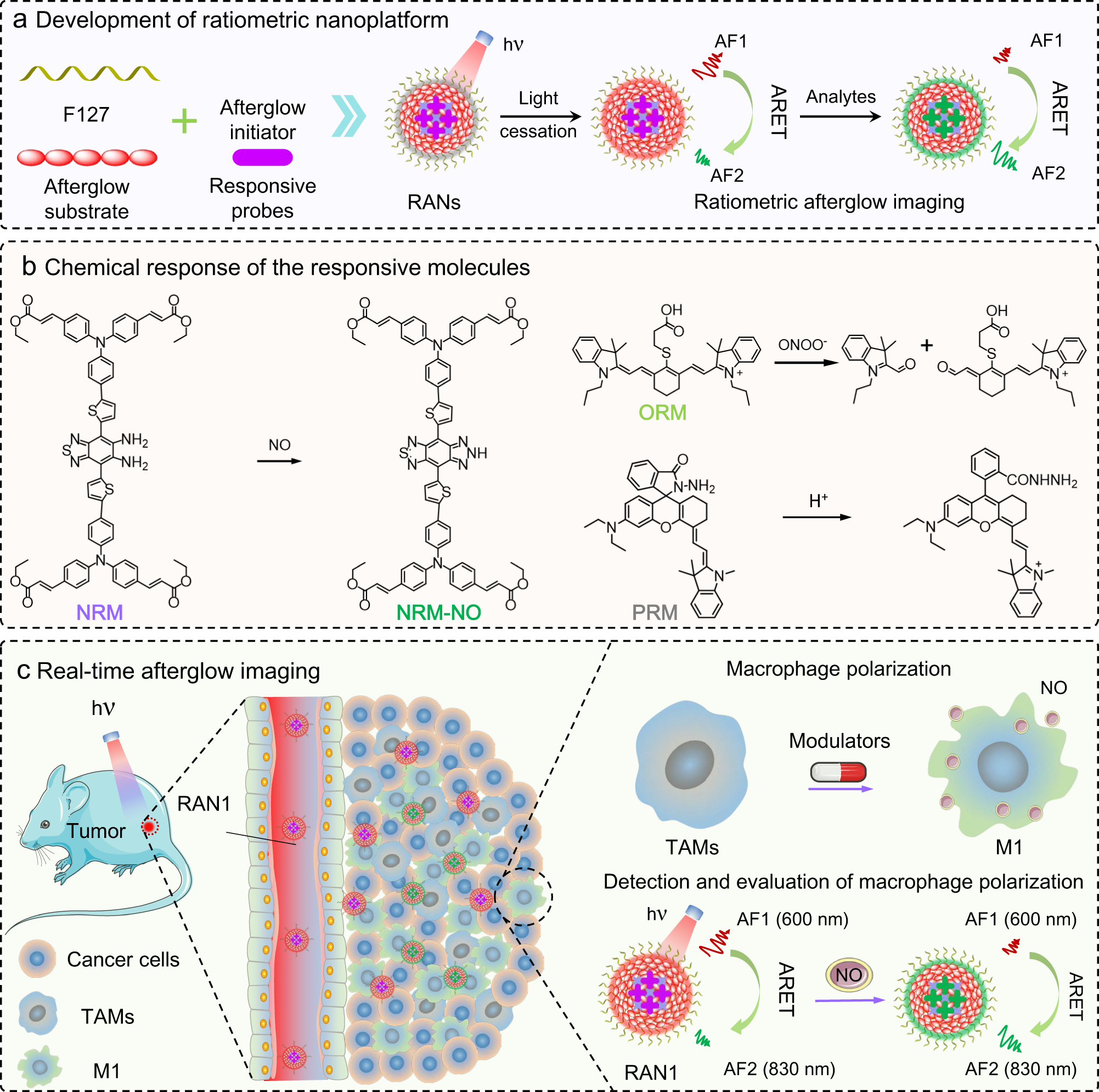
Ratiometric afterglow luminescent nanoplatform enables reliable quantification and molecular imaging

Peroxynitrite Activatable NIR-II Fluorescent Molecular Probe for Drug-Induced Hepatotoxicity Monitoring
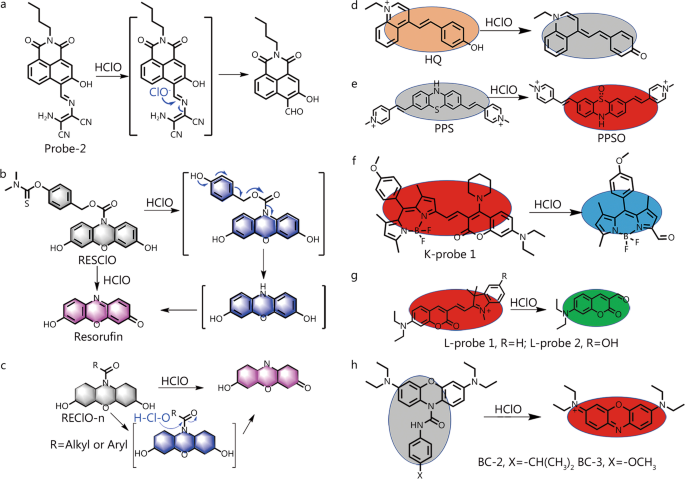
Activatable fluorescent probes for imaging and diagnosis of rheumatoid arthritis, Military Medical Research
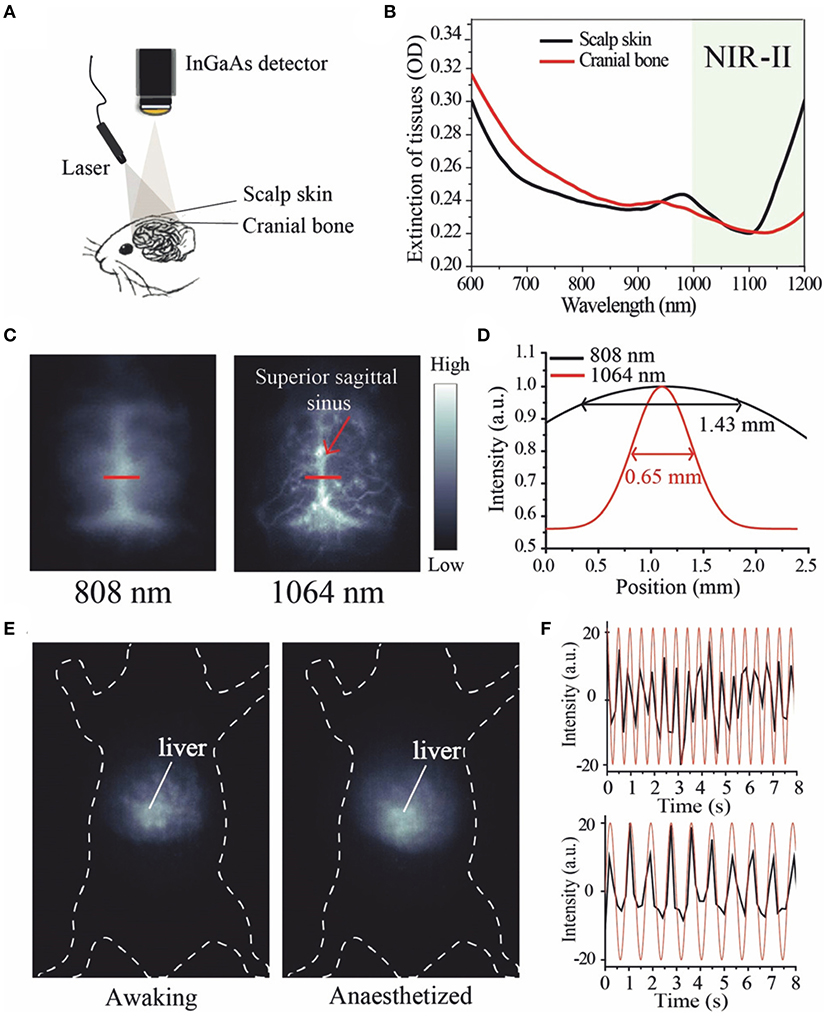
Frontiers Recent Progress in NIR-II Contrast Agent for Biological Imaging

Flexible Designing Strategy to Construct Activatable NIR-II Fluorescent Probes with Emission Maxima beyond 1200 nm

Structures and spectroscopic properties of LAP probes. (A) Design and

Bioactive NIR-II gold clusters for three-dimensional imaging and acute inflammation inhibition









