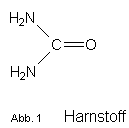CAS#:20160-71-8 N-Trimethylsilylmethyl-N'.N'-diphenyl-harnstoff
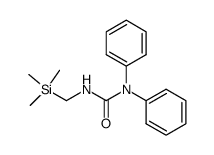
N,O-Bis(trimethylsilyl)trifluoroacetamide with trimethylchlorosilane (CAS 25561-30-2)
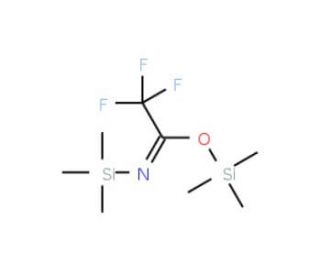
N,O-Bis(trimethylsilyl)trifluoroacetamide with trimethylchlorosilane | CAS 25561-30-2

Trimethylsilyl)methacrylate 98 13688-56-7

CAS#:20160-71-8, N-Trimethylsilylmethyl-N'.N'-diphenyl-harnstoff
Trimethylsilanol sodium salt. Sodium trimethylsilanolate is used as a catalyst in the: Synthesis of the rhodium silonate complex. Silylation of methylphenylsilane with tert-butyldimethylsilanol to synthesize trisiloxanes. Sodium trimethylsilanolate is a versatile and very powerful reagent for the conversion of esters to carboxylic acids and the hydrolysis of nitriles to primary amides.

Sodium trimethylsilanolate, CAS 18027-10-6, 95%

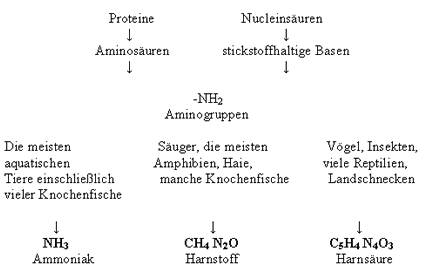
1,3-Diphenylurea 98 102-07-8

N,N-Dimethyltrimethylsilylamine 97 2083-91-2

1,1,3,3-Tetramethyl-1,3-diphenyldisilazane 96 3449-26-1
Forms azomethine ylides which readily undergo [3+2] cycloaddition to α,ß-unsaturated esters affording N-benzyl substituted pyrrolidines in good yields.

N-(Methoxymethyl)-N-(trimethylsilylmethyl)benzylamine, CAS 93102-05-7, 96%

FD162318, 943757-71-9