Auditory streaming emerges from fast excitation and slow delayed
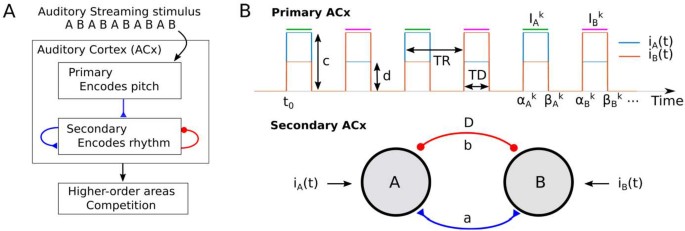
In the auditory streaming paradigm, alternating sequences of pure tones can be perceived as a single galloping rhythm (integration) or as two sequences with separated low and high tones (segregation). Although studied for decades, the neural mechanisms underlining this perceptual grouping of sound remains a mystery. With the aim of identifying a plausible minimal neural circuit that captures this phenomenon, we propose a firing rate model with two periodically forced neural populations coupled by fast direct excitation and slow delayed inhibition. By analyzing the model in a non-smooth, slow-fast regime we analytically prove the existence of a rich repertoire of dynamical states and of their parameter dependent transitions. We impose plausible parameter restrictions and link all states with perceptual interpretations. Regions of stimulus parameters occupied by states linked with each percept match those found in behavioural experiments. Our model suggests that slow inhibition masks the perception of subsequent tones during segregation (forward masking), whereas fast excitation enables integration for large pitch differences between the two tones.
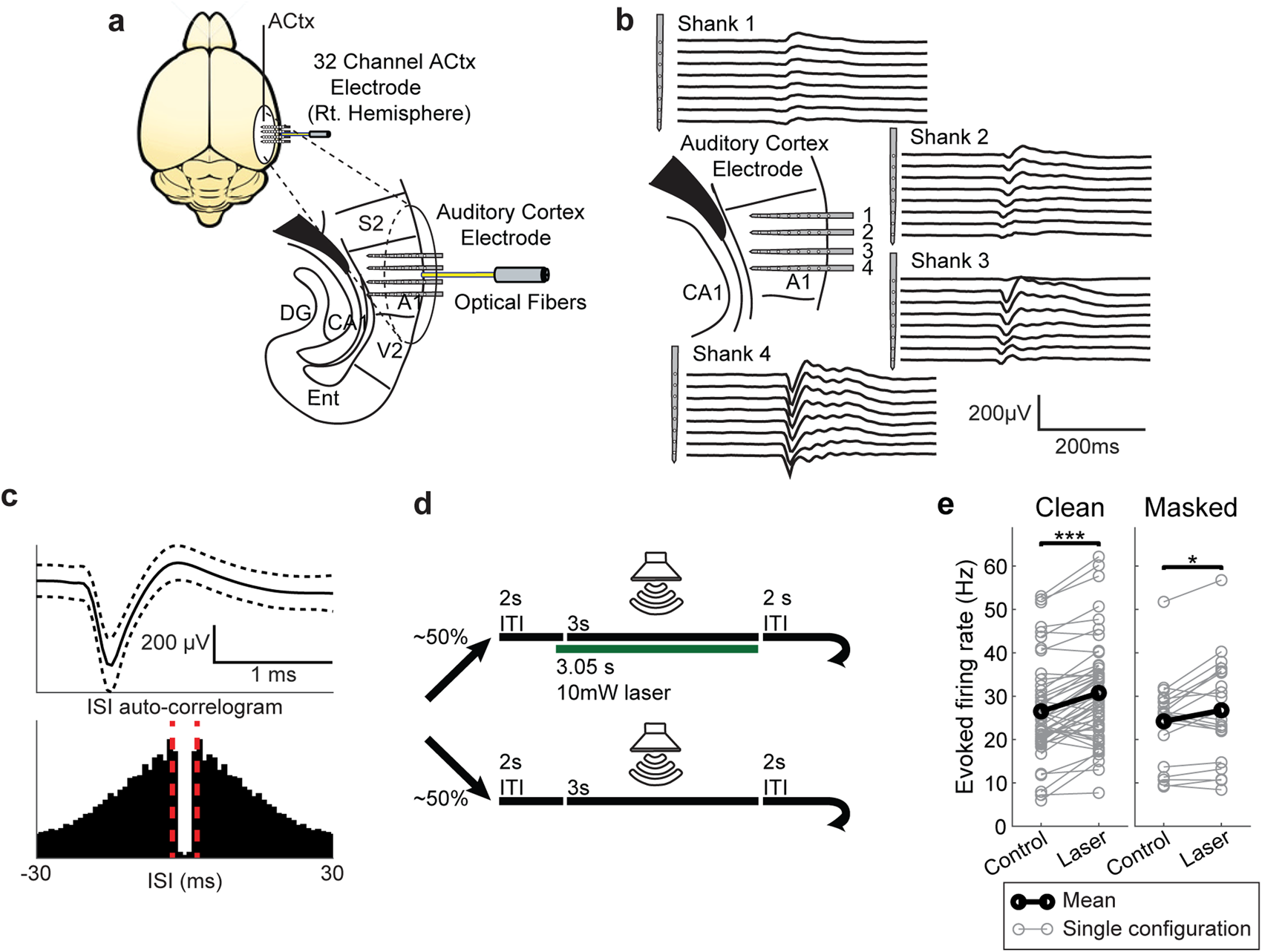
Parvalbumin neurons enhance temporal coding and reduce cortical

A) Detailed bifurcation diagram of the reduced Wilson model
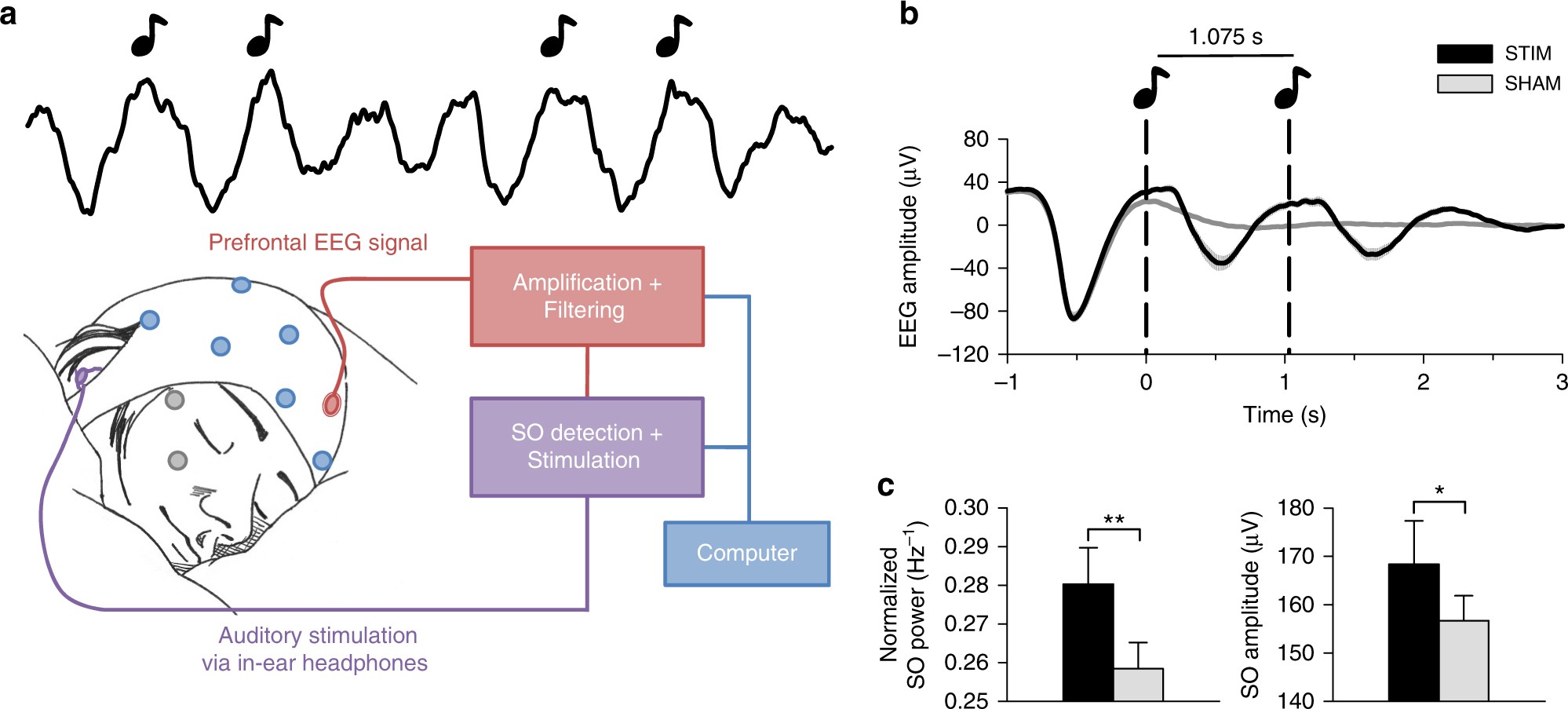
Auditory closed-loop stimulation of EEG slow oscillations

Electrical signaling in cochlear efferents is driven by an
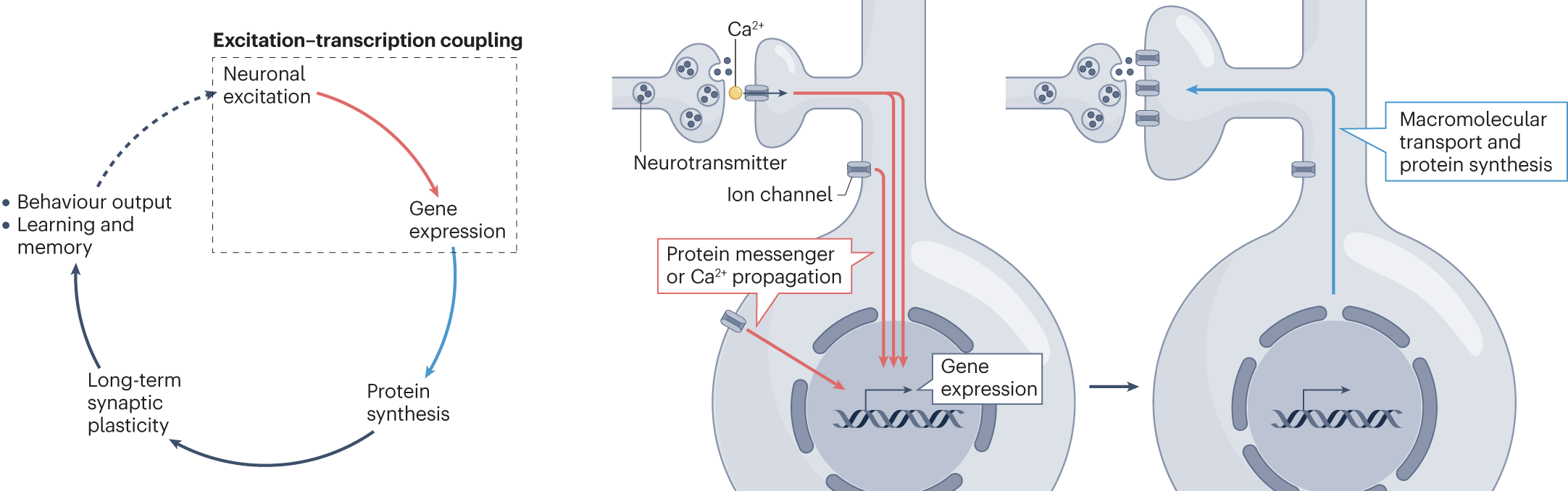
Excitation–transcription coupling, neuronal gene expression and

Auditory streaming emerges from fast excitation and slow delayed

Homeostatic Control of Spontaneous Activity in the Developing
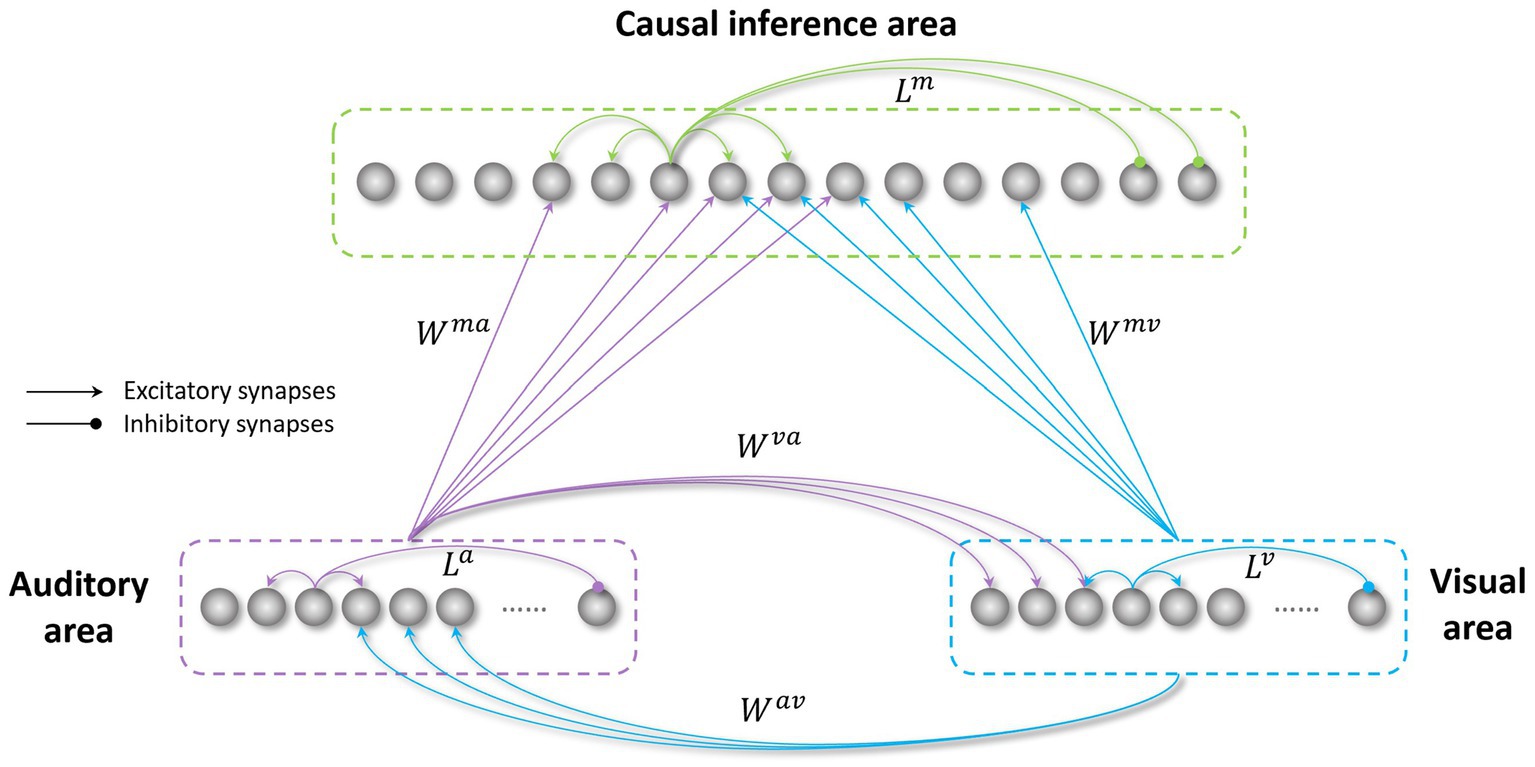
Frontiers Atypical development of causal inference in autism
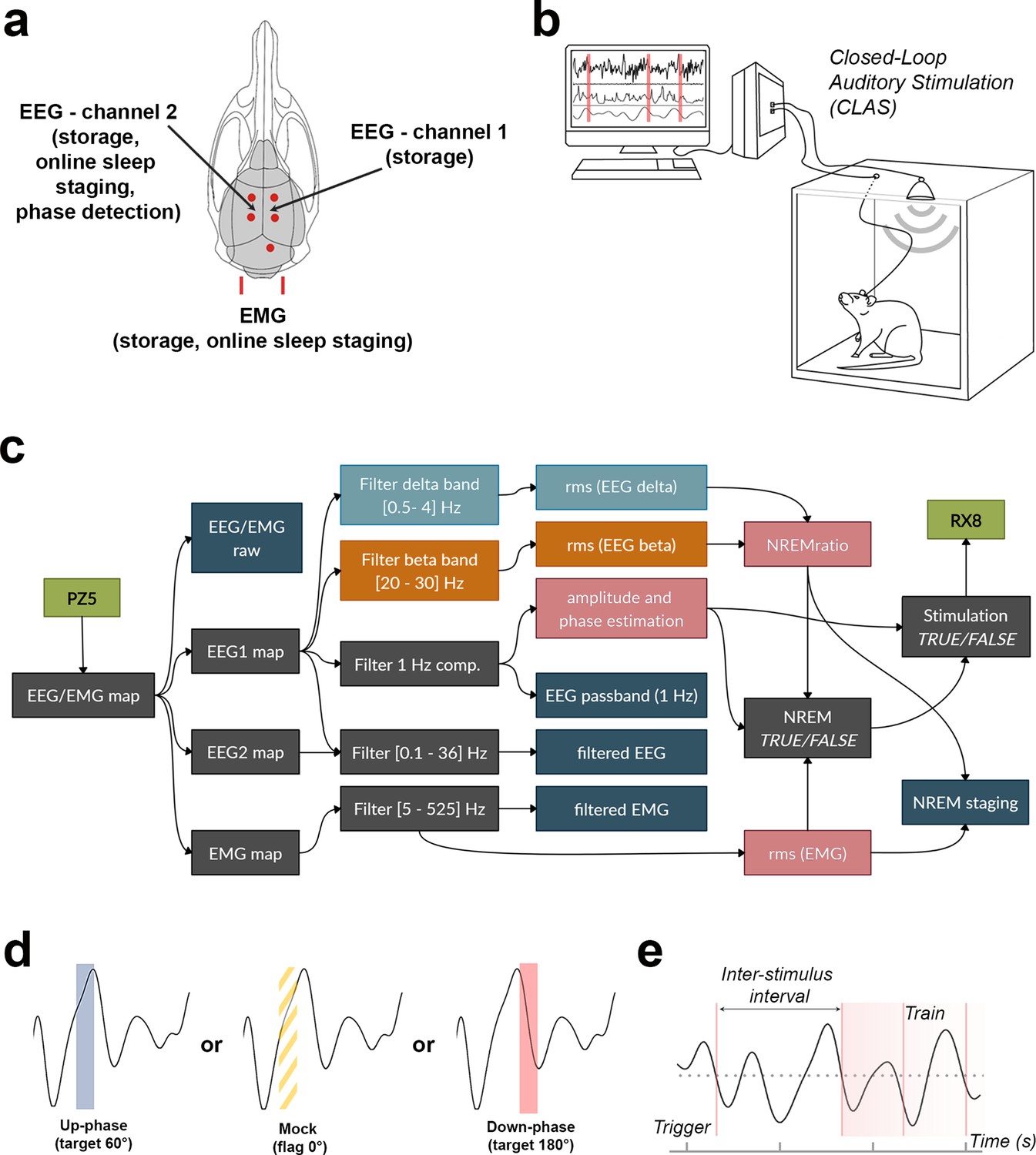
Closed-loop auditory stimulation method to modulate sleep slow

Turning the Stimulus On and Off Changes the Direction of α

Dynamic L4 survival analysis for pooled hypothesis-naive subject